   
| Debit Card The eflex Card works a like a credit card, except it has a stored value instead of a credit limit. The stored value is the employee’s available balance under the eflexFSA. The eflex Card is valid from year-to-year and automatically renewed as long as the employee is enrolled in the FSA plan up to the expiration date printed on the card. • Using the card is easy, and using your 'flex dollars' is instant. • In many cases, we don't need to ask for a receipt for purchases made using the eflex Card. • Many employers see plan participation double when they offer the eflex Card. • There are no transaction fees or pin numbers with the eflex Card. • Employees simply swipe the eflex Card at the provider location and choose the “credit card” option. • Funds are instantly withdrawn from the FSA and paid to the provider. • No manual claim filing and no more waiting for reimbursement. The card is merchant coded. Every merchant who accepts VISA is assigned one of roughly 1,000 merchant codes. The codes are listed according to industry (e.g., restaurant, pharmacy, dental office). 
NCFlex ***Please do not submit receipts for debit card transactions, unless you receive a request from P&A to do so.*** When using your NCFlex debit card, you will be. Ameriflex - Participant Services Support Center| Ameriflex Participant Services| Participant Services is available Monday through Friday from 8:30am to 8pm Eastern time at 888-868-3539. MyAmeriflex forms. To access account balance and transaction information, please log in to the myameriflex portal. Members can also submit claims and update personal. Otc card activation. Posted on February 3, 2016 by admin. Medicare replacement. Our solutions include: MyAmeriflex card (formerly Ameriflex Convenience Card). Only valid merchant codes are “open” to the card, which means if someone tries to use the card at a restaurant or gas station, it would be declined. It will also decline payment for non-covered expenses, like vitamins. The card is also coded for merchandise through the IIAS (Information Identification Approval System) at some locations, primarily pharmacies. If the merchant has implemented the IIAS, the item may be approved at the point of purchase. You may visit the Flexpert links on our website to see a list of IIAS merchants. If you're an employer, today to double your plan participation and increase your savings!
0 Comments
Zedload.com provides 24/7 fast download access to the most recent releases. We currently have 385,789 full downloads including categories such as: software, movies, games, tv, adult movies, music, ebooks, apps and much more. Our members download database is updated on a daily basis. Take advantage of our limited time offer and gain access to unlimited downloads for FREE! 
That's how much we trust our unbeatable service. This special offer gives you full member access to our downloads. Click to the Zedload today for more information and further details to see what we have to offer. Found 7 results for 3ds Max 7 Activation Code. Full version downloads available, all hosted on high speed servers! 25083 records. 3d Studio Max 7 crack 3d Studio Max 7.0 keygen Discreet 3d Studio Max 7 serials generator Discreet 3d Studio Max 7.0.3 serial key gen Electric Rain Swift 3d Max For 3ds Max 2009 serial 3d Canvas 7.1.1.2 key code generator 3d Max 8 serial keygen Milkshape 3d 1.7.10 key generator Zoom Player. 6 enter 3ds max 2010 serial number and product key. 6 enter 3ds max 2010 serial number and product key. Autodesk 3ds max 2015 product key, crack free download. Tag: Autodesk 3DS Max 2016 Crack + keygen Activation Code. No posts to display.   
Download things, please. All files are checked for viruses and clean. 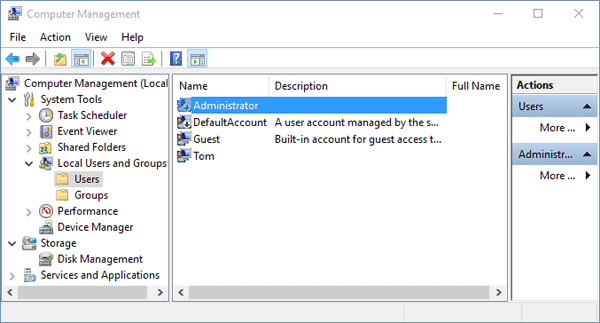
Forgot your Windows user password or accidentally delete or disable your account? Can't log into your computer because your user profile is corrupt? If it happens to be the only account with the administrative rights, you'll be locked out of the system. Fortunately, there is a built-in administrator account you can use to log on Windows. This account is the first account created during Windows installation. It has no password and is hidden by default. The account cannot be deleted or locked out. 
In this instructable i will show you guys how one can enable Administrator Account in Windows 7 The administrator account is a hidden, disabled and built-in. How to create a new user account in Windows 7 and. Name and login into their profile so they can. Enable and disable the Windows Administrator. It is a member of the Administrators group and cannot be removed from that group. Tips: In Windows XP, you can log into the built-in Administrator in Safe Mode only. However, starting from Windows Vista, the built-in administrator account is disabled by default and you can't even access it from Safe Mode. If you can still log on Windows as administrator, you can easily. Locked out of Windows and can't log on with the built-in administrator account in Safe Mode? With PCUnlocker Live CD you can enable the built-in administrator account in Windows 10, 8, 7, Vista and XP. This tool can also reset the built-in administrator password if you had changed the password but cannot remember it any longer. 13 related questions. How to Enable or Disable the Built-in Administrator Account in Windows 7. Profile of Built-in Administrator. Enable the Built-in Administrator. How to Enable the Hidden Windows 7 Admin Account Using the. To enable the hidden administrator account from within Windows. Has Viewed Your Facebook Profile? 1Create A PCUnlocker Live CD/USB Drive • to download and save the PCUnlocker self-extracting Zip file to a folder on your hard drive, then extract the ISO image file: pcunlocker.iso from it. • Insert a blank CD or USB flash drive into your computer. Download and install the freeware. 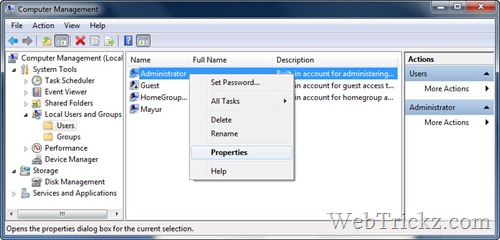
• Start the ISO2Disc program. Click the Browse button to locate your ISO image file, then click the Start Burn button to make a PCUnlocker Live CD or USB drive. 2Boot Your Locked PC from CD/USB Drive • Connect the burned CD or USB drive to your locked computer. When it is powered on, check the boot-screen for setup key (i.e. DELETE key, F8 key, F2 key) to enter BIOS Setup Utility. • Use the arrow keys to select the BOOT tab, then change the Boot Device Priority: Set 1st Boot Device to CDROM if you just created a bootable CD or DVD, or set 1st Boot Device to Removable Device if a bootable USB flash drive was created. • Press F10 to save your changes. Now your computer will restart and then boot from PCUnlocker Live CD/USB drive. If you still can't get your computer to boot from the CD/USB drive, please refer to this. 3Enable the Built-in Administrator Account • After the computer boots up from the CD or USB drive, it will start the PCUnlocker program which displays a list of local accounts that are stored in Windows SAM registry hive. Select the built-in administrator account and click on Reset Password button. • Click Yes to confirm your operation. The program will enable the built-in administrator account and also remove its password if it is protected with a password. • Click Restart to reboot the computer and remove the CD or USB drive. You can then log into Windows built-in administrator account without a password. After logging back into Windows, you can create a new administrator account for your daily work. For security purpose, it is advisable to disable the built-in administrator account again, or rename this built-in administrator account and to assign it a strong password. Warning: The built-in Administrator account has a lot more privileges than a regular administrator account—privileges that can easily get you into trouble if you use it regularly. We recommend only enabling the built-in Administrator account if you are certain you need it to troubleshoot a specific problem and then disabling it when you are done. If you’re unsure whether you need it, you probably shouldn’t use it at all. Enable Built-in Administrator Account in Windows First you’ll need to by right-clicking and choosing “Run as administrator” (or use the Ctrl+Shift+Enter shortcut from the search box). Note that this works the same in all versions of Windows. Just search for cmd and then right-click on the command prompt icon in the Start menu or Start screen. If you are in Windows 8.x or 10 you can right-click on the Start button and choose to open a command prompt that way. Now type the following command: net user administrator /active:yes. You should see a message that the command completed successfully. Log out, and you’ll now see the Administrator account as a choice. (Note that this screenshot is from Vista, but this works on Windows 7 and Windows 8 and Windows 10) You’ll note that there’s no password for this account, so if you want to leave it enabled you should change the password. Disable Built-in Administrator Account Make sure you are logged on as your regular user account, and then open an administrator mode command prompt as above. Type the following command: net user administrator /active:no The administrator account will now be disabled, and shouldn’t show up on the login screen anymore. The Amazon Fire TV and Fire TV Stick has a default screensaver with beautiful scenic photos. How to change the default screensaver on the Fire TV or Fire TV Stick. 
Description: Enables desktop screen savers. If you disable this setting, screen savers do not run. Also, this setting disables the Screen Saver section of the Screen Saver dialog in the Personalization or Display Control Panel. As a result, users cannot change the screen saver options. If you do not configure it, this setting has no effect on the system. If you enable it, a screen saver runs, provided the following two conditions hold: First, a valid screen saver on the client is specified through the 'Screen Saver executable name' setting or through Control Panel on the client computer. Second, the screen saver timeout is set to a nonzero value through the setting or Control Panel. Also, see the 'Prevent changing Screen Saver' setting. Supported on: At least Windows 2000 Service Pack 1. Microsoft official disclaimer Warning Serious problems might occur if you modify the registry incorrectly by using Registry Editor or by using another method. These problems might require that you reinstall the operating system. Microsoft cannot guarantee that these problems can be solved. Modify the registry at your own risk. Note: This registry key is created by Group Policy when this GPO is Enable or Disable. The GPO Default state is Not Configured - > this registry entry is not present. Please navigate to HKEY_CURRENT_USER Software Policies Microsoft Windows Control Panel Desktop and locate ScreenSaveActive registry key. Please select, right and copy a registry key from below, then right click on command prompt window, select Paste and press Enter Enabled: REG add 'HKCU Software Policies Microsoft Windows Control Panel Desktop' /v ScreenSaveActive /t REG_SZ /d 1 /f Disabled: REG add 'HKCU Software Policies Microsoft Windows Control Panel Desktop' /v ScreenSaveActive /t REG_SZ /d 0 /f Not Configured: REG DELETE 'HKCU Software Policies Microsoft Windows Control Panel Desktop' /v ScreenSaveActive /f Note: Manual editing of this registry key will not be reflected in Group Policy. If you modify this GPO from Group Policy this registry key will be rewritten.  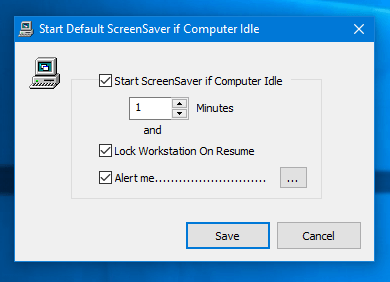  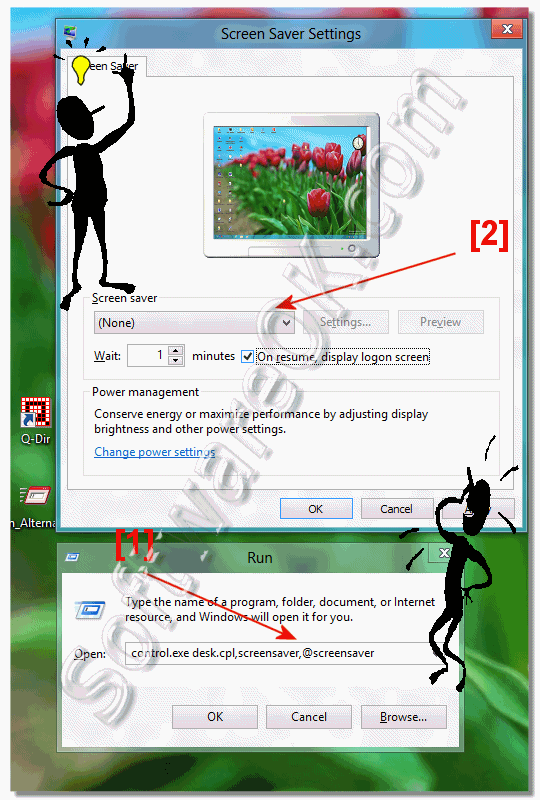 Tissue-type plasminogen activator complex Generic Name: Thrombolytic Agents This monograph includes information on the following: 1) Alteplase, Recombinant 2) Anistreplase 3) Streptokinase 4) Urokinase VA CLASSIFICATION Primary: BL115 Commonly used brand name(s): Abbokinase 4; Abbokinase Open-Cath 4; Activase 1; Activase rt-PA 1; Cathflo Activase 1; Eminase 2; Streptase 3. Other commonly used names for —Anistreplase are anisoylated plasminogen-streptokinase activator complex and APSAC. Other commonly used names for —Alteplase, Recombinant, are tissue-type plasminogen activator (recombinant), t-PA, and rt-PA. Note: For a listing of dosage forms and brand names by country availability, see Dosage Forms section(s). Category: Thrombolytic— Indications Note: Bracketed information in the Indications section refers to uses that are not included in U.S. Product labeling. General considerations The selection of thrombolytic therapy must be evaluated individually for each patient based on confirmation of thrombotic disease and assessment of patient condition and history. Some of the indications for thrombolytic therapy are identical to those for heparin or coumarin- or indanedione-derivative anticoagulants. However, the goals of thrombolytic therapy and anticoagulant therapy are different. Thrombolytic agents are used primarily to lyse obstructive thrombi and restore blood flow in a recently occluded blood vessel, whereas anticoagulants are used primarily to prevent thrombus formation and extension of existing thrombi. The potential benefit of thrombolytic therapy must be weighed against the risk of bleeding because the risk of hemorrhage may be greater with thrombolytic agents than with heparin or coumarin- or indanedione-derivative anticoagulants. Note: During its inspections of the facilities involved in the manufacture and production of urokinase, the Food and Drug Administration (FDA) identified numerous significant deviations from the Current Good Manufacturing Practice regulations; the FDA recommended that urokinase be reserved for only those situations in which a physician has determined that the use of urokinase is critical to the care of a specific patient in a specific situation. Accepted Thrombosis, coronary arterial, acute (treatment)—Alteplase, anistreplase, streptokinase, and urokinase are indicated to lyse acute coronary arterial thrombi associated with evolving transmural myocardial infarction. Alteplase and anistreplase are indicated for use via intravenous infusion; streptokinase is indicated for use via intravenous and intracoronary infusion; and urokinase is indicated for use only via intracoronary infusion.  
Various studies with intracoronary arterial injection have reported recanalization rates of 72 to 96%. In patients who received urokinase by intracoronary infusion within 6 hours following the onset of symptoms of a transmural myocardial infarction, 60% of the occluded coronary vessels were opened. However, it has not been established that the administration of urokinase during an evolving transmural myocardial infarction results in a preservation of myocardial tissue or a reduction in mortality. Additionally, intracoronary arterial administration requires prior identification of the site of the thrombus by coronary angiography. Intravenous infusion does not require coronary angiography and is the preferred route of administration because therapy can be instituted more rapidly and can be initiated in locations that lack facilities for cardiac catheterization. —Thrombolytic therapy, via intravenous or intracoronary routes of administration, may relieve chest pain, reduce the incidence of congestive heart failure, improve left ventricular function, limit cardiac damage (i.e., infarct size), and decrease the risk of early death if coronary arterial blood flow is restored before irreversible cardiac damage occurs. The reperfusion rate is dependent on the interval between the onset of symptoms and the initiation of therapy. Higher reperfusion rates are achieved when thrombolytic therapy is started within 4 hours after symptoms of ischemia first appear. However, reductions in mortality can be achieved if thrombolytic therapy is started up to 24 to 36 hours after the onset of symptoms. —Thrombolytic therapy is not a substitute for other measures that may be required to treat acute myocardial infarction or prevent reinfarction.  
Restoration of coronary arterial blood flow via thrombolysis does not correct underlying conditions that may promote thrombus formation. Recurrent ischemia, with or without reocclusion or overt reinfarction, may occur following initially successful thrombolysis. The risk of reocclusion may depend on the extent of residual stenosis in the affected vessel. Following successful thrombolytic therapy, long-term anticoagulation, platelet aggregation inhibitor therapy, percutaneous transluminal coronary angioplasty (PTCA), or coronary artery bypass graft (CABG) surgery may be required to provide long-lasting protection against reocclusion. However, initial thrombolytic therapy may permit a revascularization procedure to be performed on a delayed or elective, rather than on an emergency, basis. Stroke, acute ischemic (treatment)— Alteplase is indicated for the management of acute ischemic stroke in adults; it is used to improve neurologic recovery and reduce the incidence of disability. However, the safety and efficacy of alteplase therapy in patients with minor neurologic deficit, or with rapidly improving symptoms prior to the initiation of treatment, have not been evaluated. Thromboembolism, pulmonary, acute (treatment)— Alteplase, streptokinase, and urokinase are indicated, and may be the therapy of choice in selected patients, for the lysis of acute, massive pulmonary emboli producing obstruction or significant filling defects involving two or more lobar pulmonary arteries or an equivalent degree of obstruction in other pulmonary vessels. 
Alteplase (Activase; Cathflo Activase; TPA) is an injectable drug used to treat heart attacks, strokes. Known as tissue plasminogen activator (TPA). Medication Treatment with Alteplase IV r-tPA. Considered the gold standard,tissue plasminogen activator (or Alteplase IV r-tPA) is the only FDA-approved treatment for ischemic strokes. When promptly administered, it can save lives and reduce the long-term effects of stroke. Alteplase IV r-tPA is given through an IV in the. These agents are also indicated for lysing pulmonary emboli accompanied by unstable hemodynamics, i.e., failure to maintain blood pressure without supportive measures. Heparin is recommended for the treatment of subacute or small emboli; however, some clinicians recommend thrombolytic therapy for comparatively small emboli in patients with limited cardiopulmonary reserve caused by significant cardiac or pulmonary disease. Prior to administration of a thrombolytic agent, the diagnosis should be confirmed by objective means such as pulmonary angiography via an upper extremity vein (preferred) or ventilation-perfusion lung scanning. Thrombosis, deep venous (treatment)—Streptokinase and [urokinase ] are indicated for the lysis of acute, extensive, deep venous thrombi in the popliteal or more proximal vessels. Thrombolytic therapy may be the treatment of choice for deep venous thrombosis in selected patients. [These agents are also used for the lysis of acute, extensive thrombi in the axillary subclavian veins and vena cavae in selected patients.] However, anticoagulants are recommended for treatment of calf-vein thrombi. Prior to administration of a thrombolytic agent, the diagnosis should be confirmed, preferably by ascending venography or by Doppler ultrasound. Thromboembolism, arterial, acute (treatment) and Thrombosis, arterial, acute (treatment)—Streptokinase and [urokinase] are indicated for use via intravenous infusion for the lysis of acute arterial thrombi or emboli. [These agents are also administered locally (via a catheter positioned adjacent to or inserted into the substance of the thrombus as shown by arteriogram) to lyse arterial thrombi or emboli.] Studies have shown that thrombolytic therapy alone may be ineffective for treating chronic arterial occlusion. Angioplasty or distal bypass may be required following initial thrombolytic therapy in order to salvage the affected limb. —Streptokinase has been used in the treatment (lysis) of arterial occlusions in pediatric patients from younger than 1 month of age up to 16 years of age; however, the evidence of clinical benefits and risks in these patients is based solely on anecdotal reports. Adverse events associated with the use of streptokinase in the pediatric population are similar in nature to those associated with its use in adults, including bleeding at catheter sites. Cannula, arteriovenous, clearance—Streptokinase and [urokinase] are indicated to clear totally or partially occluded arteriovenous cannulae, as an alternative to surgical revision, when acceptable flow cannot be achieved by conventional mechanical measures. Catheter, intravenous, clearance— Alteplase and urokinase are indicated to restore patency to intravenous catheters, including central venous catheters, obstructed by clotted blood or fibrin deposits. —[Thrombolytic agents are also used to treat renal artery thrombosis, retinal blood vessel occlusions, hemolytic uremic syndrome, and impending renal cortical necrosis. However, controlled studies are required to establish the safety and effectiveness of such therapy in these conditions.] Unaccepted Thrombolytic agents should not be used to treat superficial thrombophlebitis. Alteplase has not been sufficiently studied, and is currently not recommended, for treatment of deep venous thrombosis or arterial thrombosis not associated with evolving acute myocardial infarction or for clearing occluded arteriovenous cannulae or obstructed intravenous catheters. Alteplase and streptokinase are not recommended for treatment of arterial emboli originating in the left side of the heart (e.g., mitral stenosis accompanied by atrial fibrillation) because of the risk of cerebral embolism. Streptokinase is not indicated for restoration of patency of intravenous catheters. 1 Not included in Canadian product labeling. Pharmacology/Pharmacokinetics Physicochemical characteristics: Source— Alteplase: Glycoprotein enzyme (serine protease) containing 527 amino acids; produced by recombinant DNA technology utilizing the complementary DNA (cDNA) for natural human tissue-type plasminogen activator obtained from a human melanoma cell line. Anistreplase: P-anisoylated derivative of a fibrinolytic enzyme (protein) complex consisting of human plasma-derived lys-plasminogen and bacterially derived streptokinase. Streptokinase: Co-enzyme (protein) obtained from cultures of group C, beta-hemolytic streptococci. Urokinase: Enzyme (protein) obtained from cultures of primary human neonatal kidney cells. Molecular weight— Alteplase: About 68,000 daltons Anistreplase: About 131,000 daltons Streptokinase: About 46,000 daltons Urokinase: About 33,000 daltons Mechanism of action/Effect: Thrombolytic agents activate the endogenous fibrinolytic system by cleaving the arginine 560–valine 561 bond in plasminogen to produce plasmin, an enzyme that degrades fibrin clots, fibrinogen, and other plasma proteins, including the procoagulant factors V and VIII. Alteplase and urokinase cleave the peptide bond directly. Anistreplase and streptokinase act indirectly; they combine with plasminogen to form streptokinase-plasminogen complexes that are converted to streptokinase-plasmin complexes. These activator complexes, rather than streptokinase itself, convert residual plasminogen to plasmin. These complexes are inactivated, in part, by antistreptococcal antibodies. Conversion of plasminogen to plasmin occurs within the thrombus or embolus as well as on its surface and in circulating blood. Thrombolytic agents lyse fibrin deposits wherever they exist and can be reached by the plasmin generated; therefore, thrombolytic agents also promote lysis of fibrin deposits responsible for hemostasis. Alteplase is more clot-selective than the other thrombolytic agents, binding more readily to the fibrin-plasminogen complex within a clot than to circulating (free) plasminogen. However, limited systemic fibrinolysis does occur with usual therapeutic doses. Other actions/effects: Fibrinogenolysis and fibrinolysis induced by thrombolytic agents increase the concentration of fibrinogen-degradation and fibrin-degradation products (FDP/fdp) in the blood. The FDP/fdp exert an anticoagulant effect, probably by impairing fibrin polymerization and possibly by decreasing thrombin generation and/or interfering with platelet function. Alteplase usually reduces the circulating fibrinogen concentration and increases FDP/fdp concentrations to a lesser extent than does streptokinase, but to about the same extent that urokinase does. However, studies have not shown a significantly lower incidence of bleeding with alteplase than has been reported with the other thrombolytic agents, probably because factors other than the concentrations of fibrinogen and/or FDP/fdp also significantly influence the risk of bleeding (see ). Specifically, the risk of bleeding complications associated with thrombolytic therapy may be more dependent on the presence of vascular injury than on the extent of systemic fibrinolysis induced by a specific agent. Anistreplase has potent proteolytic activity in the systemic circulation. In addition to decreasing plasma concentrations of fibrinogen, the medication lowers plasma concentrations of plasminogen, procoagulant factors V and VIII, and the fibrinolysis inhibitor alpha-2-antiplasmin. Anistreplase, streptokinase, and urokinase have also been reported to decrease plasma viscosity and erythrocyte aggregation, probably as a result of reduced fibrinogen concentration. Streptokinase and the streptokinase component of anistreplase are antigenic and induce the formation of antibodies. Elevation of the antistreptokinase antibody titer usually occurs about 5 to 7 days following administration, reaches a peak after 2 to 3 weeks, and may persist for 1 year or longer. The antibodies may cause resistance to subsequent streptokinase or anistreplase therapy, and possibly an increased risk of anaphylaxis or other severe allergic reactions. Biotransformation: Alteplase—Hepatic; rapid. Streptokinase—Hepatic; no metabolites identified. Urokinase—Hepatic; rapid. Half-life: Alteplase: Distribution: Less than 5 minutes. Elimination: Approximately 35 minutes. Anistreplase: The half-life of anistreplase's fibrinolytic activity is 70 to 120 minutes (average about 90 minutes ). The deacylation half-life of the complex is about 105 to 120 minutes. The plasma clearance and duration of fibrinolytic activity of the medication are probably controlled primarily by its deacylation rate. Streptokinase: Following intravenous administration of 1.5 million International Units (IU) over a 1-hour period: the half-life of the activator complexes (streptokinase-plasminogen and/or streptokinase-plasmin) is 23 minutes. Urokinase: Up to 20 minutes. The half-life may be prolonged in patients with hepatic function impairment. Time to peak effect: Reperfusion of the myocardium generally occurs 20 minutes to 2 hours (average 45 minutes) following initiation of intravenous therapy. Duration of action: Thrombolysis may continue for approximately 4 hours following administration of alteplase, streptokinase, or urokinase; the hyperfibrinolytic effect disappears within a few hours following discontinuation of administration. Following administration of anistreplase, thrombolysis may continue for approximately 6 hours, and a systemic hyperfibrinolytic state, as demonstrated by euglobulin clot lysis time determinations, may persist for more than 2 days. For all thrombolytic agents, the prothrombin time may rarely be prolonged for 12 to 24 hours following cessation of therapy because of the decreased plasma concentration of fibrinogen, decreased plasma concentration of factor V and possibly other coagulant factors, and/or the anticoagulant effects of FDP/fdp. However, prolonged, high FDP/fdp concentrations may potentiate bleeding for a longer period of time, especially after administration of non–clot-selective thrombolytic agents. Elimination: Alteplase—Renal; approximately 80% of a dose is excreted in the urine, as metabolites, within 18 hours. Urokinase—Small quantities are eliminated via the renal and biliary routes. Precautions to Consider Cross-sensitivity and/or related problems Patients allergic to streptokinase will be allergic to anistreplase also, and vice versa. Carcinogenicity Alteplase, anistreplase, and urokinase—Long-term studies to determine whether alteplase, anistreplase, and urokinase have carcinogenic potential have not been done. Mutagenicity Alteplase—No mutagenicity was demonstrated in the Ames test or in chromosomal aberration assays in human lymphocytes. Anistreplase—No mutagenicity was demonstrated in chromosomal aberration assays in human lymphocytes. Pregnancy/Reproduction Fertility— Alteplase: Studies have not been done in animals. Anistreplase: Studies have not been done in humans. Studies have not been done in animals. Urokinase: Studies in mice and rats have not shown that urokinase causes impaired fertility. Pregnancy— It has been suggested that administration of a thrombolytic agent during the first 18 weeks of pregnancy may increase the risk of premature separation of the placenta because fetal attachments to the uterus during this time are composed primarily of fibrin. However, this problem has not been reported following administration of streptokinase or urokinase to patients during the first 2 trimesters of pregnancy. Alteplase and anistreplase Studies have not been done in humans. Studies have not been done in animals. FDA Pregnancy Category C. Streptokinase Streptokinase apparently crosses the human placenta minimally if at all. However, antibodies to streptokinase do cross the placenta. Studies in pregnant women (treated mostly during the second and third trimesters) have not shown evidence of abnormalities or induction of fibrinolysis in the fetus. Studies have not been done in animals. FDA Pregnancy Category C. Urokinase Adequate and well-controlled studies have not been done in humans. Studies in mice and rats have not shown that urokinase causes fetal harm when administered in doses up to 1000 times the human dose. FDA Pregnancy Category B. Postpartum — Thrombolytic agents should be administered with great caution during the first 10 days postpartum because of the increased risk of hemorrhage. Breast-feeding It is not known whether thrombolytic agents are distributed into breast milk. However, problems in humans have not been documented. Pediatrics Safety and efficacy have not been established. Although controlled clinical studies have not been conducted to determine the safety and efficacy of using streptokinase in pediatric patients, a significant number of anectodal reports exist supporting the use of streptokinase in children, particularly for the treatment of arterial occlusions. In patients from younger than 1 month of age up to 16 years of age, the use of streptokinase for the treatment of acute arterial occlusions resulted in bleeding complications in as many as 50% of catheter sites in some studies. Occasionally, bleeding has required blood transfusion. Careful monitoring of the patient is therefore recommended. Geriatrics Geriatric patients generally have a poorer prognosis than younger adults following an acute myocardial infarction. Also, they may be more likely than younger adults to have pre-existing conditions that tend to increase the risk of intracranial bleeding or other hemorrhagic complications. Because the risks of thrombolytic therapy, as well as its potential benefits, are increased in older patients, careful patient selection and monitoring are recommended. Drug interactions and/or related problems The following drug interactions and/or related problems have been selected on the basis of their potential clinical significance (possible mechanism in parentheses where appropriate)—not necessarily inclusive ( » = major clinical significance): Note: Combinations containing any of the following medications, depending on the amount present, also may interact with this medication. In addition to the interactions listed below, the possibility should be considered that multiple effects leading to further impairment of blood clotting and/or increased risk of hemorrhage may occur if a thrombolytic agent is administered to a patient receiving any medication having a significant potential for causing hypoprothrombinemia, thrombocytopenia, or gastrointestinal ulceration or hemorrhage. ALTEPLASE, RECOMBINANT Summary of Differences Indications: Indicated in the treatment of acute coronary arterial thrombosis, acute ischemic stroke, acute pulmonary thromboembolism, and in the clearance of central intravenous catheters. Pharmacology/pharmacokinetics: Mechanism of action/effect— Acts directly to convert plasminogen to plasmin. May be more clot-selective than anistreplase, streptokinase, or urokinase. Half-life— Biphasic; about 4 minutes for distribution phase and 35 minutes for elimination phase. Side/adverse effects: Incidence of stroke and cerebral hemorrhage greater than with other thrombolytic agents. Incidence and severity of allergic reactions lower than with anistreplase or streptokinase. Additional Dosing Information Alteplase is not antigenic (as are anistreplase and streptokinase) and does not promote antibody formation. Therefore, a second course of alteplase therapy can be administered, if reocclusion occurs, without resistance having developed to the effects of alteplase and without risk of precipitating an anaphylactic reaction. In one study, a second course of alteplase therapy was shown to be effective, without producing significant bleeding complications, in patients exhibiting signs and symptoms of reocclusion following initial thrombolytic therapy for treatment of acute myocardial infarction. However, it must still be considered that a second course of therapy, if initiated before systemic effects of the first dose have subsided, may increase the risk of severe hemorrhage. A large multi-center study has shown that alteplase, administered in an accelerated or front-loaded dosing regimen within 6 hours of the onset of symptoms of myocardial infarction, may achieve earlier and more complete patency of the infarct-affected artery than does streptokinase in combination with intravenous or subcutaneous heparin, or a combination of alteplase, streptokinase, and intravenous heparin. Twenty-four-hour and 30-day mortality was also lower with the accelerated or front-loaded alteplase regimen than with these combinations. For treatment of acute ischemic stroke The treatment of acute ischemic stroke with alteplase should be limited to facilities that can provide appropriate evaluation and management of intracranial hemorrhage. Treatment should be initiated only within 3 hours after the onset of stroke symptoms, and after exclusion of intracranial hemorrhage by a cranial computed tomographic scan or other diagnostic imaging method sensitive to the presence of hemorrhage. In patients who have not recently been treated with oral anticoagulants or heparin, alteplase may be given prior to the availability of coagulation study results. However, treatment should be stopped if either a pre-treatment prothrombin time > 15 seconds or an elevated APTT is identified. Blood pressure should be monitored frequently and controlled with appropriate medication during and following alteplase administration. The safety and efficacy of concomitant administration of heparin and aspirin during the first 24 hours after symptom onset have not been investigated. Parenteral Dosage Forms ALTEPLASE, RECOMBINANT, FOR INJECTION Usual adult dose Intravenous catheter clearance After the intravenous tubing has been disconnected and catheter occlusion confirmed, the catheter should be filled with a solution containing 1 mg per mL of alteplase. The solution should be administered as follows: Initial Dose • For patients weighing 30 kg or more—Intravenous, 2 mg in 2 mL. • For patients weighing at least 10 kg and less than 30 kg—Intravenous, 110% of internal lumen volume of the catheter, not to exceed 2 mg in 2 mL. Second dose • If catheter function is not restored at 120 minutes after dose 1, a second dose may be administered.. Note: There is no efficacy or safety information on dosing in excess of 2 mg per dose for this indication. Studies have not been performed with administration of total doses greater than 4 mg (two 2 mg doses). Thrombosis, coronary arterial, acute Standard regimen: For patients weighing less than 65 kg—Intravenous, 1.25 mg per kg of body weight administered over a period of three hours, as follows: • First hour—60% of the total dose. Initially, 6 to 10% of the total dose is given by direct intravenous injection within the first one or two minutes. The next 50 to 54% of the total dose is given via intravenous infusion during the remainder of the hour. • Second hour—20% of the total dose, via intravenous infusion. • Third hour—20% of the total dose, via intravenous infusion. For patients weighing 65 kg or more—Intravenous, 100 mg, administered over a period of three hours, as follows: • First hour—60 mg. Initially, 6 to 10 mg is given by direct intravenous injection within the first one or two minutes. The next 50 to 54 mg is given via intravenous infusion during the remainder of the hour. • Second hour—20 mg, via intravenous infusion. • Third hour—20 mg, via intravenous infusion. Accelerated regimen: For patients weighing less than 67 kg—Intravenous, initially 15 mg followed by an infusion of 0.75 mg per kg of body weight, up to 50 mg, administered over a period of thirty minutes. The infusion should continue for an additional sixty minutes at a dose of 0.5 mg per kg of body weight, up to 35 mg. For patients weighing 67 kg or more—Intravenous, initially 15 mg by direct intravenous injection, followed by 50 mg infused over the next thirty minutes, and then 35 mg infused over the next sixty minutes. Note: It is recommended that intravenous heparin be administered in conjunction with accelerated-dose alteplase at an initial dose of 5000 USP Heparin Units, followed by 1000 USP Heparin Units per hour (1200 USP Heparin Units per hour for patients weighing more than 80 kg), with the dose adjusted to raise the activated partial thromboplastin time to between sixty and eighty-five seconds. Stroke, acute ischemic Intravenous, 0.9 mg per kg of body weight (up to a maximum of 90 mg) infused over sixty minutes, with 10% of the total dose administered by direct intravenous injection over the first minute. Thromboembolism, pulmonary, acute Intravenous infusion, 100 mg, administered over a period of two hours. Note: It is recommended that heparin be used in conjunction with alteplase for treatment of acute pulmonary embolism. Heparin should be administered only if the patient's activated partial thromboplastin time or thrombin time value is no higher than twice the control value, near the end of or immediately following the alteplase infusion. Usual pediatric dose Safety and efficacy have not been established. ANISTREPLASE Summary of Differences Indications: Indicated in the treatment of acute coronary arterial thrombosis. Pharmacology/pharmacokinetics: Mechanism of action/effect—Acts indirectly to promote conversion of plasminogen to plasmin. Other actions/effects—Antigenic; promotes antibody formation. Half-life—70 to 120 minutes (average about 90 minutes). The deacylation half-life of the complex is about 105 to 120 minutes. Precautions: Medical considerations/contraindications—Caution is required in patients who have had a severe hypersensitivity reaction to prior anistreplase or streptokinase therapy or a prior course of anistreplase or streptokinase therapy within the past 12 months. Side/adverse effects: Incidence of mild hypersensitivity and febrile reactions greater than with alteplase or urokinase. May cause severe hypersensitivity reactions including anaphylaxis. Additional Dosing Information It is recommended that equipment and medications (such as epinephrine, glucocorticoids, and antihistamines) for treating anaphylaxis be immediately available whenever anistreplase is administered. Some investigators have administered a glucocorticoid (e.g., 100 mg of hydrocortisone or methylprednisolone, intravenously) and/or an antihistamine (e.g., 50 mg of diphenhydramine, intravenously) prior to anistreplase administration, to decrease the risk of severe hypersensitivity and febrile reactions. However, the prophylactic efficacy of these medications has not been established. Resistance to anistreplase therapy may occur because of the presence of high titers of antibodies following a prior course of anistreplase or streptokinase therapy. A significant titer of these antibodies generally occurs 5 to 7 days following administration of anistreplase or streptokinase and may persist for 1 year (up to 4 years in some patients). Alteplase or urokinase may be administered if thrombolytic therapy is indicated during this time. A recent streptococcal infection also may result in high titers of antibodies and resistance to anistreplase. Parenteral Dosage Forms ANISTREPLASE FOR INJECTION Usual adult dose Thrombosis, coronary arterial, acute Intravenous, 30 units, administered over two to five minutes. Usual pediatric dose Safety and efficacy have not been established. Size(s) usually available: U.S.— 30 Units per single-dose vial (Rx) [ Eminase ( human albumin 30 mg) (dimethylsulfoxide. STREPTOKINASE Summary of Differences Indications: Indicated in the treatment of acute coronary arterial thrombosis, acute pulmonary thromboembolism, deep venous thrombosis, and acute arterial thromboembolism and thrombosis. Also indicated to clear totally or partially occluded arteriovenous cannulae. Not indicated for restoration of patency of intravenous catheters. Pharmacology/pharmacokinetics: Mechanism of action/effect—Acts indirectly to promote conversion of plasminogen to plasmin. Other actions/effects—Antigenic; promotes antibody formation. Half-life—Following rapid, high-dose administration: 23 minutes (as active activator complex activity). Precautions: Medical considerations/contraindications—Caution required in patients who have had a severe hypersensitivity reaction to prior streptokinase therapy or a prior course of streptokinase therapy within the past 12 months. Side/adverse effects: Incidence of mild hypersensitivity and febrile reactions greater than with urokinase or alteplase. May cause severe hypersensitivity reactions including anaphylaxis or, rarely, skin lesions. Additional Dosing Information It is recommended that equipment and medications (such as epinephrine, glucocorticoids, and antihistamines) for treating anaphylaxis be immediately available whenever streptokinase is administered. Some investigators have administered a glucocorticoid (e.g., 100 mg of hydrocortisone or methylprednisolone, intravenously) and/or an antihistamine (e.g., 50 mg of diphenhydramine, intravenously) prior to streptokinase administration, to decrease the risk of severe hypersensitivity and febrile reactions. However, the prophylactic efficacy of these medications has not been established. Resistance to streptokinase therapy may occur because of the presence of high titers of antibodies to streptokinase following a prior course of streptokinase or anistreplase therapy. A significant titer of these antibodies generally occurs 5 to 7 days following administration of anistreplase or streptokinase and may persist for 1 year (up to 4 years in some patients). Alteplase or urokinase may be administered if thrombolytic therapy is indicated during this time. A recent streptococcal infection also may result in high titers of antibodies and resistance to streptokinase. For intravenous administration of streptokinase (for indications other than acute coronary arterial thrombosis), a loading dose of 250,000 International Units (IU) is recommended to overcome mild resistance caused by exposure (without recent active infection) to streptococci. Since this loading dose successfully overcomes resistance in 85 to 90% of patients, many clinicians state that a previously recommended resistance test is now considered unnecessary. However, if a thrombin time (TT) determination or other test of fibrinolysis performed after 4 hours of therapy indicates minimal or no fibrinolytic activity, and no clinical improvement is apparent, the possibility of excessive resistance to streptokinase should be considered. Streptokinase should be discontinued and an alternate thrombolytic agent (alteplase or urokinase, but not anistreplase) administered instead. A previously recommended regimen of variable maintenance dosage with frequent laboratory monitoring has not been shown to increase the efficacy or safety of streptokinase therapy. Therefore, this regimen is not currently recommended and has been replaced by a fixed maintenance dosage schedule. The dosage and duration of intravenous therapy vary with the condition being treated. For the individual patient, tests to determine restoration of blood flow, such as angiography or venography of the affected blood vessel, computed tomography, impedance plethysmography, or quantitative Doppler effect, may be useful in determining the optimum duration of administration. Parenteral Dosage Forms STREPTOKINASE FOR INJECTION Usual adult dose Thrombosis, coronary arterial, acute Intravenous, 1,500,000 IU, administered within one hour. Intra-arterial (via a coronary artery catheter placed via the Judkins or Sones technique), 20,000 IU initially, followed by 2000 IU per minute for one hour. Note: Recanalization may occur in less than one hour; however, treatment should be continued following recanalization, to ensure complete lysis of all thrombotic material. The solution may be used for intravenous administration within 8 hours following reconstitution when stored between 2-30°C (36-86°F). Do not add other medication to solutions containing Activase. Any unused solution should be discarded. During and following Activase administration for the treatment of AIS, frequently monitor and control blood pressure. In patients without recent use of oral anticoagulants or heparin, Activase treatment can be initiated prior to the availability of coagulation study results. Discontinue Activase if the pretreatment International Normalized Ratio (INR) is greater than 1.7 or the activated partial thromboplastin time (aPTT) is elevated. Contraindications Do not administer Activase to treat acute ischemic stroke in the following situations in which the risk of bleeding is greater than the potential benefit: current intracranial hemorrhage (ICH); subarachnoid hemorrhage; active internal bleeding; recent (within 3 months) intracranial or intraspinal surgery or serious head trauma; presence of intracranial conditions that may increase the risk of bleeding; bleeding diathesis; and current severe uncontrolled hypertension. Do not administer Activase to treat acute myocardial infarction or pulmonary embolism in the following situations in which the risk of bleeding is greater than the potential benefit: active internal bleeding; history of recent stroke; recent (within 3 months) intracranial or intraspinal surgery or serious head trauma; presence of intracranial conditions that may increase the risk of bleeding; bleeding diathesis; and current severe uncontrolled hypertension. Warnings and Precautions Bleeding Activase can cause significant, sometimes fatal, internal or external bleeding, especially at arterial and venous puncture sites. Avoid intramuscular injections and trauma to the patient. Fatal cases of hemorrhage associated with traumatic intubation in patients administered Activase have been reported. The concomitant administration of heparin and aspirin with and following infusions of Activase during the first 24 hours after symptom onset has not been investigated. Because heparin, aspirin, or Activase may cause bleeding complications, carefully monitor for bleeding, especially at arterial puncture sites. Hemorrhage can occur 1 or more days after administration of Activase, while patients are still receiving anticoagulant therapy. If serious bleeding occurs, terminate the Activase infusion. References 1. National Institute of Neurological Disorders and Stroke. Know the signs. National Institute of Neurological Disorders and Stroke website. Last modified March 29, 2016. Accessed August 12, 2016. Jauch EC, Saver JL, Adams HP Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Demaerschalk BM, Kleindorfer DO, Adeoye OM, et al. Scientific Rationale for the Inclusion and Exclusion Criteria for Intravenous Alteplase in Acute Ischemic Stroke: A Statement for Healthcare Professionals From the American Heart Association/American Stroke Association. FDA Information on Medication Errors Involving Activase and TNKase. September 2015. Accessed August 9, 2016. Activase [Prescribing Information]. South San Francisco, CA: Genentech, Inc; January 2017. National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. FDA Access Summary basis of approval. Accessed August 14, 2017. Stroke Reperfusion Therapy: IV t-PA Administration. Massachusetts General Hospital: Stroke Services Web Site. Last updated: January 16, 2015. Accessed September 29, 2017. 9. Summers D, Leonard A, Wentworth D, et al. Comprehensive overview of nursing and interdisciplinary care of the acute ischemic stroke patient: a scientific statement from the American Heart Association. American Association of Neurosicence Nurses (AANN) Reference Series for Clinical Practice. Guide to the Care of the Patient with Ischemic Stroke. Accessed August 9, 2016. Tamm A, Siddiqui M, Shuaib A, et al. Impact of stroke care unit on patient outcomes in a community hospital. CMS Quality Measurement Programs Characteristics. Centers for Medicare and Medicaid Services website. Published June 28, 2012. Accessed August 13, 2016. The Joint Commission, Stroke. Last updated June 17, 2016. Accessed August 1, 2016. Busby L, Owada K, Dhungana S, et al. CODE FAST: a quality improvement initiative to reduce door-to-needle times. J Neurointerv Surg. Jauch EC, Cucchiara B, Adeoye O, et al. Part 11: adult stroke: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. 2010;122(18 Suppl 3):S818-S828. TARGET: STROKE PHASE II, Campaign Manual. Accessed August 2, 2016. Target: StrokeSM Phase II. 11 key best practice strategies. October 2014. Accessed March 30, 2015. Kalanithi L, Tai W, Conley J, Platchek T, Zulman D, Milstein A. Better health, less spending: delivery innovation for ischemic cerebrovascular disease. American Hospital Association. October 2012. Accessed October 11, 2016. Major pulmonary embolism: review of a pathophysiologic approach to the golden hour of hemodynamically significant pulmonary embolism. Tan KT, Oudkerk M, van Beek EJR. Deep Vein Thrombosis and Pulmonary Embolism. In: Hallet JW Jr, Mills JL, Earnshaw JJ, Reekers JA, Eds. Comprehensive Vascular and Endovascular Surgery. Philadelphia, PA: Elsevier Limited; 2003. Kearon C, Akl EA, Ornelas J, Blaivas A, et al. Antithrombotic Therapy for VTE Disease: CHEST Guideline and Expert Panel Report. 2016 Feb;149(2):315-52. Fesmire FM, Brown MD, Espinosa JA, et al. Critical issues in the evaluation and management of adult patients presenting to the emergency department with suspected pulmonary embolism. Ann Emerg Med. 2011 Jun;57(6):628-652.e75. Skolarus LE, Meurer WJ, Shanmugasundaram K, et al. Marked regional variation in acute stroke treatment among Medicare beneficiaries. Accessed August 14, 2017. Follow these steps for reconstituting 50-mg vials. 5 • Reconstitute Activase immediately before administration, using only preservative free Sterile Water for Injection (SWFI), U.S. Pharmacopeia (USP). • A single 50-mg vial of Activase may be used to treat patients weighing up to 122 lbs (55.5 kg). Reconstitute Activase immediately before administration, using aseptic technique at all times. • Begin by assembling the 50-mg vial of Activase, the vial of SWFI USP, (included with the Activase package), a 50-mL syringe, a large-bore needle (eg, 18-gauge), and alcohol wipes. Also, prepare a polyvinyl chloride bag or glass vial along with an infusion set. • Reconstitution should be carried out by adding SWFI to the 50-mg vial of Activase powder. Step 1: Remove the protective cap from the top of the Activase vial and the vial of SWFI. Swab the top of each vial with an alcohol wipe to reduce the risk of contamination. Step 2: Using the large-bore needle and syringe, withdraw 50 mL of SWFI. Do not use Bacteriostatic Water for Injection, USP. Do not prime the syringe with air. Safely discard the diluent vial according to institutional procedures. Step 3: Insert the syringe into the stopper on the 50-mg vial of Activase and inject the contents, directing the stream into the lyophilized cake. DO NOT USE IF VACUUM IS NOT PRESENT. Safely discard the needle and syringe according to institutional procedures. Step 4: Mix the solution with a gentle swirl. DO NOT SHAKE. Slight foaming of the solution is normal. Let the solution stand undisturbed for several minutes to allow any large bubbles to dissipate. Visually inspect the Activase solution for particulate matter and discoloration before administration. Get the latest news, updates, and information about our product You are now signing up for information that is intended for US healthcare professionals only. By signing up at this site, you certify that you are a healthcare professional licensed in the United States or its territories and are indicating that you wish to receive information about our product, other Genentech products, and related disease education. Genentech's intent is to only provide information to healthcare professionals licensed in the United States or its territories who would likely be treating patients within the FDA-approved indications for this product. Select • Critical Care Medicine (Emergency Medicine) • Emergency Medicine Hospitalist • Neurological Surgery • Neurology • Neurology/Diagnostic • Neuroradiology • Radiology By completing and submitting this form, you agree to allow Genentech and its agents to collect the information provided and to be contacted by Genentech and its agents in the future regarding our products, other Genentech products, and related disease education. Genentech will not sell, rent or otherwise distribute your name and any personally identifiable information outside of Genentech and its agents. Genentech will only use your information in accordance with its. 
• Live View shooting does not work in Basic Zone modes. • During Live View shooting, do not point the camera toward the sun. The sun's heat can damage the camera's internal components. • Live View shooting will disable the following Custom Function settings;[C.Fn III 8 Mirror lockup] [C.Fn IV 9 Shutter/AE lock button][C.Fn IV 10 SET button when shooting]. • Under low light or bright light conditions, the Live View image might not reflect the brightness of the captured image. • If the light source within the image changes, the screen might flicker. 
If this happens, stop the Live View shooting, then resume Live View shooting under the actual light source to be used. • During Live View image display, if you point the camera in a different direction, it might throw off the correct brightness momentarily and the image might not look right. Wait until the image brightness stabilizes before shooting. • If there is a very bright light source in the picture, such as the sun, the bright area might be blackened on the LCD monitor. However, the actual captured image will correctly show the bright area. • If you set the [LCD brightness] to bright settings in low light, chrominance noise may appear in the Live View image. However, the chrominance noise will not be recorded to the captured image. • When you magnify the image, the image sharpness may look more pronounced than it really is. • Under the [Custom Functions (C.Fn)] menu, setting [C.Fn II 5 Auto Lighting Optimizer] to [0: Enable] may display a darkly-exposed (set with exposure compensation) image brighter. Focusing Manually. • will be the same as using. • You can set the ISO speed, white balance, and exposure compensation during Live View shooting. • The Picture Style, drive mode, AF mode, and AF point cannot be changed during Live View shooting. • During Live View shooting, the metering mode will be set to evaluative metering regardless of the current metering mode setting. • You can check the depth of field by pressing the depth-of-field preview button. • During continuous shooting, the exposure set for the first shot will also be applied to subsequent shots. • When you use Live View shooting for a long period, the camera's internal temperature may increase and it can degrade image quality. Terminate Live View shooting when not shooting images. Before taking a long exposure, stop Live View shooting temporarily and wait several minutes before shooting. • High temperatures, high ISO speeds, or long exposures may cause noise or irregular colors in the image captured with Live View shooting. • If you take the picture while the image is magnified, the exposure might not come out as desired. Return to the full view before taking the picture. During the magnified view, the shutter speed and aperture will be displayed in red. • Even if you take the picture during magnified view, the captured image will reflect the full view. • The focus preset feature on super telephoto lenses cannot be used. • For Live View shooting, do not use an Extension Tube or TS-E lens. Doing so can cause irregular or unintended exposures. • FE lock is not possible when the built-in flash or external Speedlite is used. Modeling flash is not possible when external Speedlite is used. • A non-Canon flash will not fire. Using AF to Focus. • Low-contrast subject such as a blue sky and solid-color surfaces. • Subjects in low light. Jan 16, 2009 How to set Live View on Canon EOS Rebel XS?! Easy Steps to Use the Live View Function of EOS REBEL T1i/EOS 500D to Shoot Still Images. • Stripes and other patterns where there is contrast only in the horizontal direction. • Under a light source whose brightness, color, or pattern keeps changing. • Night scenes or points of light. • Under fluorescent lighting or when the image flickers. • Extremely small subjects. • Subjects strongly reflecting light. • When both near and far subjects are covering the AF points, such as an animal in a cage. • Subjects which keep moving within the AF point and cannot keep still due to camera shake or subject blur.  
• A subject approaching or leaving the camera. • When AF is used for an extremely out-of-focus subject. • When shooting a soft-focus image with a soft-focus lens. • When using a special effect filter. 
• Tell us some more • Upload in progress • Upload failed. Please upload a file larger than 100 x 100 pixels • We are experiencing some problems, please try again. • You can only upload files of type PNG, JPG or JPEG. How do you activate blackberry messenger? - I have already topped up £5 and all I have to do is actually activate BBM, but I can't seem to find out how to do that. Nov 23, 2017. Learn how to make a video call and how much it will cost you. • You can only upload files of type 3GP, 3GPP, MP4, MOV, AVI, MPG, MPEG or RM. • You can only upload photos smaller than 5 MB. • You can only upload videos smaller than 600 MB. • You can only upload a photo (png, jpg, jpeg) or video (3gp, 3gpp, mp4, mov, avi, mpg, mpeg, rm). • You can only upload a photo or video. • Video should be smaller than 600 MB/5 minutes • Photo should be smaller than 5 MB •. 
May 30, 2017. 2.2.1 How to activate; 2.2.2 How to top up. 2.3.1 How to top up. 3 VoIP applications; 4 GPS and other navigation applications. US$ 9.43 and allows you to check emails, chat with friends and families with BlackBerry Messenger (BBM) and other Instant Messaging services (Yahoo! 
Hi, I have posted the message below to the Microsoft Windows Xp email help service. I have decided to post a copy here as well so that there is more chance someone who reads it may be able to find me a solution. Orignal message follows.  
------------------------------------------ What are you trying to do? Activate Windows XP through internet 2. Use Windows Update 3. Do the above on a multi-boot PC (Win 98 + Win XP) 4. Access this section of support with XP (I am using Win 98 to access this page) How are you trying to do it? Trying to activate using the taskbar icon or start menu link. Trying to use Windows Update from internet explorer or the start menu link. Windows XP was installed sucessfully to an available 18Gb SCSI drive (E, leaving previous OS (Win 98) untouched on drive C: 4. Trying to access these support pages with Windows XP What is happening as a result? Aug 10, 2016. Try resetting your firewall service. Right-click start, control panel, Administrative services, services. Scroll down to Windows Firewall, right-click properties. Set startup type to - Automatic and click [start]. Dec 22, 2017 GridinSoft Anti-Malware 3.1.14 Final Crack+Activation Key Serial. Making Windows 10 Run Blazingly Fast For Free. Haig Dickson 32,781 views. 
I see - 'checking for connectivity.' After a timeout period of approx. 1 or 2 minutes I get - 'Unable to activate Windows Online activation cannot be completed at this time. Message number: 32781' 2. When I tried to use Windows Update, it successfully checked my version of Windows Update and installed the latest version. I can get as far as the screen which says - 'welcome to Windows update keep your computer up to date check to see if you need updates for windows, your hardware or your devices' Then if I click either of the buttons 'express' or 'custom' update, I see 'checking for updates for your computer' and animated green squares moving along a progress bar. After some time this is follow by '[Error number: 0x80072EE2] The website has encountered a problem and cannot display the page you are trying to view. The options provided below might help you solve the problem. For self-help options: etc.' There does not appear to be any problem selecting which OS to use at boot up. I do not know if using dual boot OS is causing the above 2 problems. Nothing happens for ages, then get a 'page could not be loaded' type error, as if the computer is not connected. How often is this problem happening? Every time, for all problems. Xp was installed yesterday. I now have 29 days left to find the solution. I know I can activate using the 'phone up' method, but that is hardly solving the problem which is preventing internet activation is it? Surely whatever is causing this activation problem is related to the Windows Update problem as well, and activating by phone is just a 'bandaid' solution which will not fix the underlying problem. Most web browsing seems to work, but I've noticed that I cannot sign in to google groups in order to post on newsgroups while in XP. ------------------------------------------ Has anyone experienced these problems? How can I fix them? For now I guess I am stuck with using Windows 98 for internet and XP for everything else (at least until it locks me out or I phone up the activation). If the multiboot systems are both on your C drive, you would need a different partition for each, to make them both work. Ferguson FAQ for Windows Antispy 'fred' wrote in message news:d9frkr$j8s$. > Hi, I have posted the message below to the Microsoft Windows Xp email help service. I have decided to post a copy here as well so > that there is more chance someone who reads it may be able to find me a solution. > > orignal message follows. > ------------------------------------------ > What are you trying to do? Activate Windows XP through internet > 2. Use Windows Update > 3. Do the above on a multi-boot PC (Win 98 + Win XP) > 4. Access this section of support with XP (I am using Win 98 to access this page) > > How are you trying to do it? Trying to activate using the taskbar icon or start menu link. Trying to use Windows Update from internet explorer or the start menu link. Windows XP was installed sucessfully to an available 18Gb SCSI drive (E, leaving previous OS (Win 98) untouched on drive C: > 4. Trying to access these support pages with Windows XP > > What is happening as a result? I see - 'checking for connectivity.' > After a timeout period of approx. 1 or 2 minutes I get - > 'Unable to activate Windows > Online activation cannot be completed at this time. > Message number: 32781' > 2. When I tried to use Windows Update, it successfully checked my version of Windows Update and installed the latest version. I > can get as far as the screen which says - 'welcome to Windows update > keep your computer up to date > check to see if you need updates for windows, your hardware or your devices' > Then if I click either of the buttons 'express' or 'custom' update, I see 'checking for updates for your computer' and animated > green squares moving along a progress bar. After some time this is follow by '[Error number: 0x80072EE2] The website has > encountered a problem and cannot display the page you are trying to view. The options provided below might help you solve the > problem. > For self-help options: etc.' There does not appear to be any problem selecting which OS to use at boot up. I do not know if using dual boot OS is causing > the above 2 problems. Nothing happens for ages, then get a 'page could not be loaded' type error, as if the computer is not connected. > > How often is this problem happening? > Every time, for all problems. > > Xp was installed yesterday. I now have 29 days left to find the solution. > I know I can activate using the 'phone up' method, but that is hardly solving the problem which is preventing internet activation > is it? Surely whatever is causing this activation problem is related to the Windows Update problem as well, and activating by > phone is just a 'bandaid' solution which will not fix the underlying problem. > Most web browsing seems to work, but I've noticed that I cannot sign in to google groups in order to post on newsgroups while in > XP. > ------------------------------------------ > > Has anyone experienced these problems? How can I fix them? For now I guess I am stuck with using Windows 98 for internet and XP > for everything else (at least until it locks me out or I phone up the activation). Ferguson wrote: > If the multiboot systems are both on your C drive, you would need a different partition for each, to make them both work. > As, I said, XP was installed to a separate drive (E. Anyway, I never recieved any response from Microsoft support, but I have somehow corrected the problem myself. I booted in Win98, then deleted all WinXP files and directories (hidden and system too)from drive E:, then I reinstalled XP on E: again, only this time all internet functionality was working correctly. I didn't do anything different this time, so I am at a loss as to why it did not work the first time I installed. Hi, If you could do that then UAC would not be a viable security measure. MSDN forum can help with programming methods of working with UAC. MSDN - Forums MSDN - Where is the Forum For? ------------------------------------------------------------------------------------------------------ Some other information that might help. How to Create a Elevated Program Shortcut without a UAC Prompt How to Change UAC Notification Settings in Windows 7 Windows 7: User Account Control (UAC) Explained (UAC Disabled method at end of article) Disable User Account Control (UAC) the Easy Way on Win 7 or Vista| Enable or Disable UAC From the Windows 7 / Vista Command Line ========================================== References to Vista also apply to Windows 7. Disable User Account Control (UAC) for certain Windows Vista applications VistaUACMaker – Make XP Based Programs and Applications Compatible With Vista and Windows 7 Ignore UAC for specific programs How to Run Programs Elevated Without Getting the UAC Prompt Selectively disable UAC for your trusted Vista applications You can also turn UAC to QUIET or OFF though this does lower your security level. THIS UTILITY MAKES IT EASY Tweak UAC Utility - put UAC in Quiet Mode or On/OFF easily Hope this helps. Rob Brown - Microsoft MVP - Windows and Devices for IT 2010 - current Windows Insider MVP 2016 - current. 
Aug 8, 2009 - 2 min - Uploaded by nerdishcoukHey, in this video ill show you How To Turn Off UAC (User Account Control) in Windows 7. Feb 16, 2014 Enable or Disable User Account Control (UAC) Prompt for Built-in Administrator in Windows Information User Account Control (UAC) helps pr.  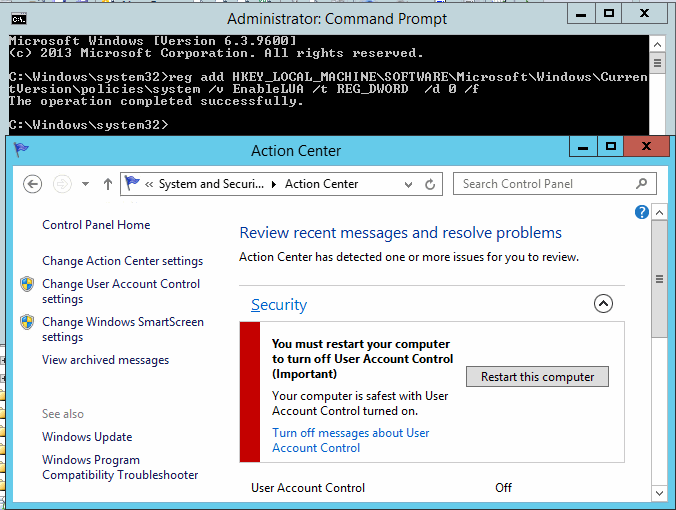
We need your advice in handling discounts configuration considering the below explained scenario: Standard SAP configuration we know of for cash discount provides two options to recognize cash discount: net or gross. The net procedure is accomplished in the system by taking the cash discount into account during document posting (this uses a discount clearing account). This means, the cash discount is immediately deducted at time of invoice so that inventory (or expense) account is reduced. If you set an item or a group of items to active, the agreed cash discount is proposed automatically. The cash discount is active if the cash discount date has not expired. The cash discount is inactive if the cash discount period has expired. You can then activate the cash discount by double-clicking the cash discount amount. Automatic Cash discount config in SAP. Automatic posting for cash discount in SAP 1. Automatic Posting for Cash Discount Granted| SCN. AN OVERVIEW DOCUMENT SPLITTING IN NEW GL IN SAP ECC IS ONE. 01100 Company code clearing 01300 Cash discount clearing. SAP allows you to activate.  
In all other transactions (gross procedure), the system posts the cash discount when you clear the open items (this uses a discount revenue account). This means, the full value of the inventory/expense remains intact (i.e. Not reduced), and the discount is posted at time of payment to a separate discount revenue/expense account. My client wants to use the net procedure but does not want to recognize the discount at time of invoice posting. They want to recognize the discount at time of payment, and the deduction should go back to the cost of the invoice Is there any way we can accomplish this in standard SAP? • Hi, Two questions about Cash Discount: 1. The Cash discount on SAP standard solution is generated from the FI module? 
In our case the invoice is created through SD and the Pricing Procedure. • SAP FI (Financial Accounting) IntroductionThe SAP FI CO (Financial Accounting & Controlling) Module has the capability of meeting all the accounting and financial needs of an organization. • T-code (OBXI) (Caution: First create a Cash Discount Allowed A/C in GL) Path Financial Accounting Accounts Receivables & Payables Business Transactions. • Configuration Document Financial Accounting (FI) 1 Financial Accounting This section covers the introduction of Financial Accounting. 1.1 Financial Accounting Global Settings Global.  |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
